In today's current global health situation, the demand for COVID-19 Antigen and Antibody test kits are seriously increasing.
To ensure the safety, quality and performance of all COVID-19 test kits available in the market, it is imperative to set standard parameters for these In Vitro Diagnostic (IVD) products.
FDA Memorandum No. 2020-011 and FDA Memorandum No. 2020-022 were issued requiring mandatory performance testing of COVID-19 antibody and antigen test kits with FDA issued Special Certification by the Research Institute for Tropical Medicine (RITM), respectively, as part of the post-marketing surveillance of COVID-19 In Vitro Diagnostics (IVDs).(1)
In fulfillment of this and to ensure access to novel quality diagnostic assays for COVID-19, RITM has standardized its evaluation protocols and endeavored to streamline the product evaluation pathway into 4 stages as described below.
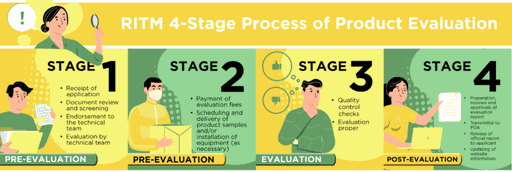
As of January 17, 2022, GENEDIA W COVID-19 Ag by Green Cross Medical Science is currently on stage 3 of RITM evaluation.
Click the link below to view more :
As the official distributor of GENEDIA W Covid-19 Ag here in the Philippines, we are confident that we will meet the specificity and sensitivity rate set by the Department of Health.
_______________________________________________________
Interested on Genedia?
Contact us.
